Test Your Periodontitis Knowledge
Let's test your periodontitis knowledge!
Research suggests there are various oral manifestations of inflammatory bowel disease (IBD) even though they are not frequently detected by general practitioners and gastroenterologists alike. Manifestations of IBD can include halitosis, dysphagia, aphthous ulcerations, deep oral fissuring, lichen planus, and more.
A systemic review conducted in 2019 searched articles in multiple databases to collect data regarding oral manifestations of inflammatory bowel disease (IBD). Since most general practitioners and gastroenterologists do not frequently detect these oral manifestations, it is imperative for dental professionals to recognize symptoms in order to allow for a multidisciplinary approach for preventing, diagnosing, and treating the disease. According to the review, the prevalence of oral manifestations of IBD ranges from 0.7% to 37% in adults and symptoms can include mucosal tags, cheilitis granulomatosa, halitosis, dysphagia, aphthous ulcerations, deep oral fissuring, lichen planus, and more.
Lauritano, D.; Boccalari, E.; Stasio, D.D.; Vella, F.D.; Carinci, F.; Lucchese, A.; Petruzzi, M. Prevalence of Oral Lesions and Correlation with Intestinal Symptoms of Inflammatory Bowel Disease: A Systematic Review. Diagnostics 2019, 9, 77. Retrieved from https://www.mdpi.com/2075-4418/9/3/77
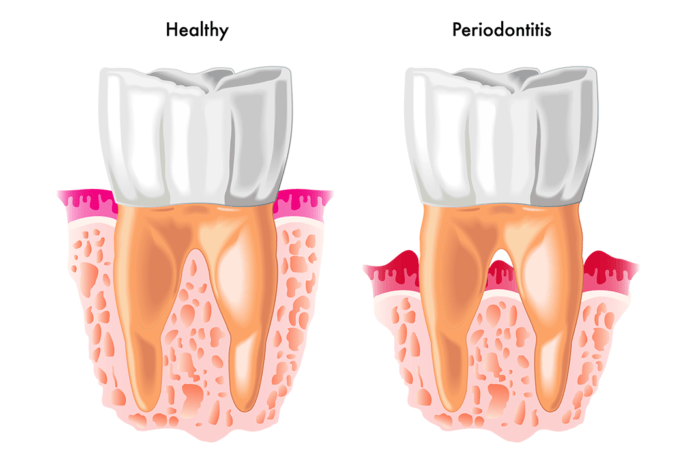
Porphyromonas gingivalis is a Gram-negative bacteria that is widely recognized as the main pathogen in chronic periodontitis and is also associated with which of the following systemic diseases?
Porphyromonas gingivalis is a Gram-negative bacteria found in the oral cavity that causes microbial dysbiosis. Widely recognized as the main pathogen in chronic periodontitis, it is anaerobic, rod-shaped, and may even have the capability to protect itself against foreign elements such as bacteriophages and viruses. P. gingivalis is also associated with multiple systemic diseases such as cardiovascular and respiratory diseases, rheumatoid arthritis, premature birth, and Alzheimer’s disease.
Chen T, Olsen I. Porphyromonas gingivalis and its CRISPR-Cas system. J Oral Microbiol. 2019;11(1):1638196. Published 2019 Jul 3. doi:10.1080/20002297.2019.1638196
According to the updated periodontitis classifications released at the 2017 World Workshop on the Classification of Periodontal and Peri-implant Diseases and Conditions, periodontitis is now identified in the following categories only:
According to the updated periodontitis classifications released at the 2017 World Workshop on the Classification of Periodontal and Peri-implant Diseases and Conditions, periodontitis classifications have been updated to align with emerging scientific evidence. The following three distinct categories are now accepted: necrotizing periodontitis, periodontitis as a manifestation of systemic disease, and periodontitis. These revised categories focus on the severity of disease, complexity of the disease management, and includes a grading system that can be adapted as new evidence emerges.
Caton, J, Armitage, G, Berglundh, T, et al. A new classification scheme for periodontal and peri‐implant diseases and conditions – Introduction and key changes from the 1999 classification. J Periodontol. 2018; 89( Suppl 1): S1– S8. https://doi.org/10.1002/JPER.18-0157
According to the 2017 proceedings of the World Workshop on the Classification of Periodontal and Peri-implant Diseases and Conditions, it is now accepted that a patient with periodontitis may revert back to health and only require intermittent treatment for periodontitis.
In 2017, a committee at the World Workshop on the Classification of Periodontal and Peri-implant Diseases and Conditions was charged with updating the 1999 classification of periodontal diseases and conditions in order to reflect current understanding and evidence. While the updated classification system remained the same for certain classifications, the committee also modified and added new categories. For instance, the workshop concluded that though a patient with gingivitis can revert to a state of health, a patient with periodontitis must remain a periodontitis patient for life. New guidelines were also created to add specific verbiage for peri-implant diseases and conditions.
Caton, J, Armitage, G, Berglundh, T, et al. A new classification scheme for periodontal and peri‐implant diseases and conditions – Introduction and key changes from the 1999 classification. J Periodontol. 2018; 89( Suppl 1): S1– S8. https://doi.org/10.1002/JPER.18-0157
After comparing plaque samples from 200 systemically and periodontally healthy smokers and non-smokers, one study concluded that the pathogen profile of smokers consisted of a highly diverse, pathogen rich, anaerobic microbiome that is similar to someone who presents with disease.
A systemic review conducted in 2019 summarized current research regarding the impact of smoking on the human microbiome and its involvement in periodontitis and other diseases. It was found that cigarette smokers have a higher risk of severe periodontitis than non-tobacco users. Current research has focused on the associations between smoking and sub-gingival bacteria and concludes that smoking-associated periodontitis contains a microbial profile that is distinct from that of non-smokers. Likewise, after comparing plaque samples from 200 systemically and periodontally healthy smokers and non-smokers, it was shown that the pathogen profile of smokers consisted of a highly diverse, pathogen rich, anaerobic microbiome that is similar to someone who presents with disease.
Huang C. and Shi, G. (2019). Smoking and microbiome in oral, airway, gut, and some systemic diseases. Journal of Translational Medicine. 17(225). Retrieved from https://translational-medicine.biomedcentral.com/articles/10.1186/s12967-019-1971-7